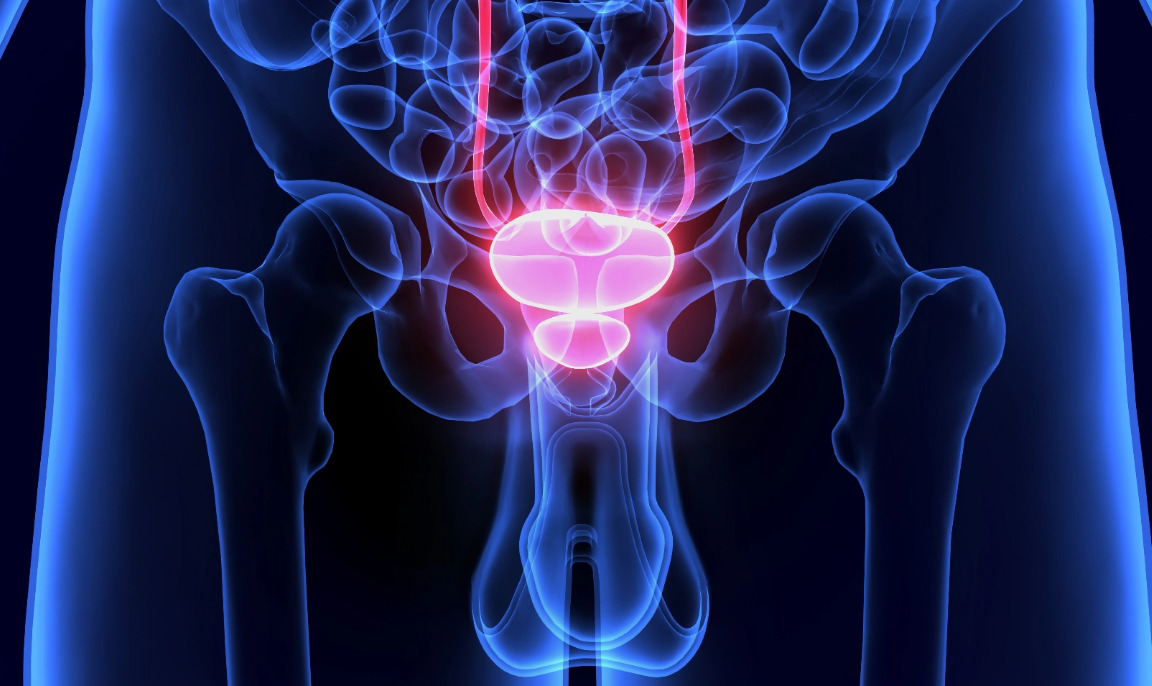
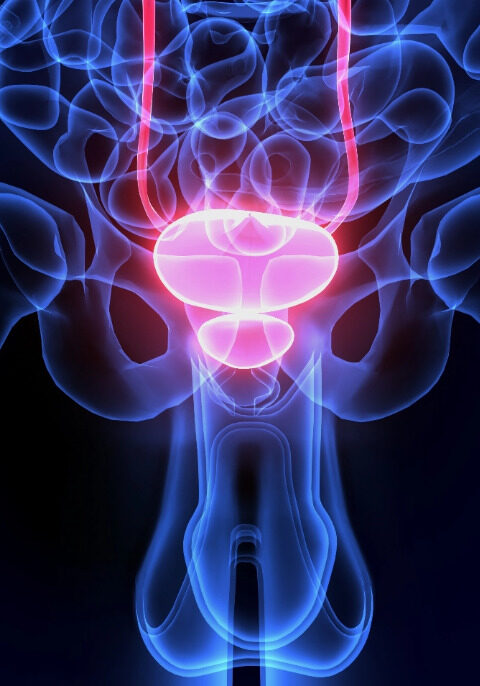
What is Focal Therapy?
What is focal therapy?
Focal therapy is a targeted treatment approach for prostate cancer that focuses on treating only the specific areas of the prostate affected by cancer, rather than the entire gland. It aims to preserve healthy tissue and minimize side effects associated with more aggressive treatments. Techniques like HIFU, NanoKnife, or laser ablation are used to precisely treat the cancer cells in the targeted region.
Focal therapy is considered for localized cases and offers a potentially less invasive treatment option while maintaining quality of life.

 Statistics
Statistics
75 to 79
most common in men
aged 75 to 79 years.
1 in 6
1 in 6 men in the UK
will get prostate cancer at
some point in their lives
98% of men had no problems with urinary continence after HIFU
Want to know more?
Our team are here to answer any questions.
Any questions?
Being diagnosed with prostate cancer can be very distressing, and it’s normal to feel anxious, fearful and unsure which treatment to choose. We understand the emotions you may be experiencing and are here to help you to make a confident, informed decision about your care.
If you've got any questions about your prostate cancer diagnosis or want to know more about HIFU or NanoKnife focal don’t hesitate to get in touch with our friendly, knowledgeable team.
Fill in an enquiry form

