World-Class Prostate Cancer Treatment to Preserve your Quality of Life

Treatments Completed

and Erectile problems
Why choose Focal Therapy?
State-of-the-art Treatment
HIFU
High-intensity focused ultrasound (HIFU) is one of two types of focal therapy we offer. It uses ultrasound waves to target cancer in the lower, or posterior, part of your prostate. HIFU is a non-invasive treatment that is guided by advanced 3D MRI imaging to destroy cancer cells while protecting healthy tissues.- Soundwave-based treatment
- Preserves urinary and sexual function
- Day procedure and rapid recovery
NanoKnife
NanoKnife is the other type of focal therapy we offer and uses electrical pulses to target the upper, or anterior, part of your prostate. Guided by 3D MRI imaging, NanoKnife is a minimally-invasive treatment that destroys cancer cells that may be hard to reach with HIFU.- Needle-based treatment
- Preserves urinary and sexual function
- Day procedure and rapid recovery
What is focal
therapy?
Focal therapy is a NICE-approved, less invasive treatment that involves removal of specific cancerous tissue, rather than the entire prostate. We offer two types of focal therapy at The Focal Therapy Clinic.
Is focal therapy
right for me?
Focal therapy offers better outcomes for some people with prostate cancer. Find out if your prostate cancer is suitable to be treated with focal therapy today.
Treatment
options
Focal therapy isn't one single treatment. Find out about the different methods of focal therapy we can offer, from HIFU to NanoKnife.
Treatment
Costs
At The Focal Therapy Clinic, we are proud to work with all major insurers to deliver focal therapy. We aim to keep Focal Therapy affordable for self-pay patients.
At one year after one focal therapy treatment
*Based upon a review of 270 patients who all had treatment at The Focal Therapy Clinic using advanced MRI-ultrasound fusion technology to ensure treatment accuracy
Watch Steve’s Story
Advocacy and campaigning
As well as delivering outstanding care, we are also committed to changing attitudes and improving practice across private and NHS practice.
We have built a network of thought leaders, campaigners and researchers, and champion patient voices as a force for change.
What our patients say
World-Leading Hospitals for Prostate Cancer Treatment
Meet our World Leading Clinical Specialists
Learn more about Focal Therapy and Prostate Cancer
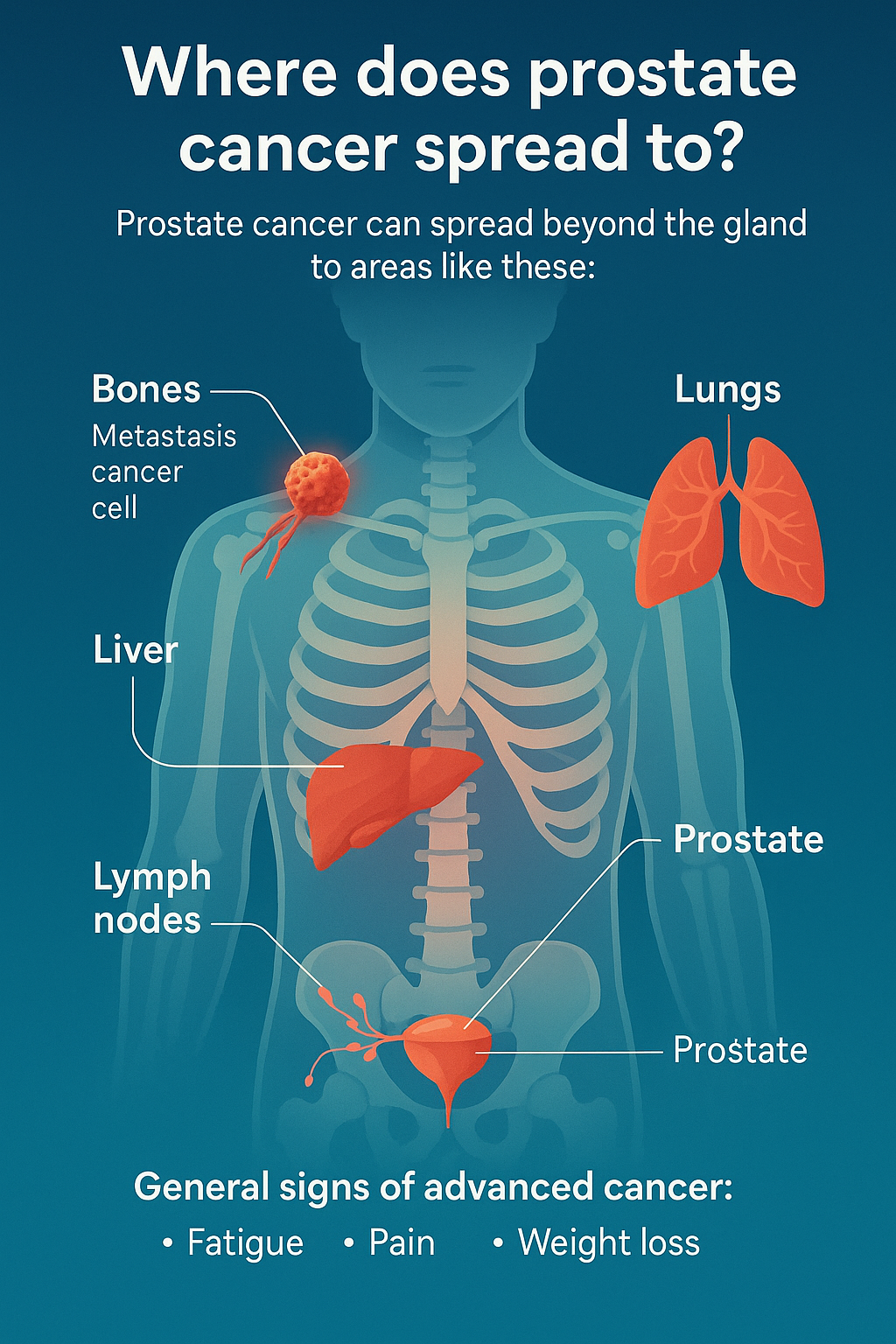
Where does prostate cancer spread to?
What is the most accurate test for prostate cancer?
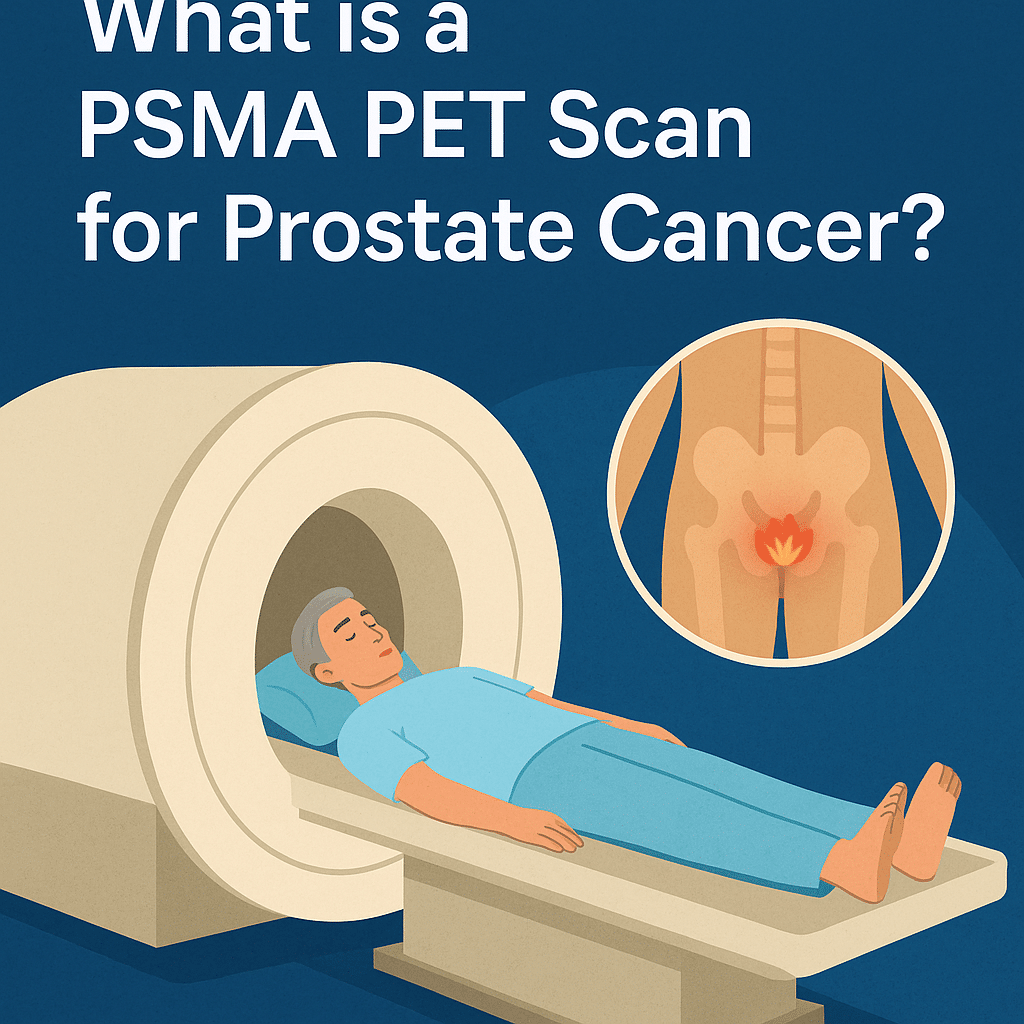
What is a PSMA PET scan for prostate cancer?
Any questions?
If you’ve got any questions about your prostate cancer diagnosis or want to know more about HIFU or NanoKnife, don’t hesitate to get in touch with our friendly, knowledgeable team.
Contact the team